Supramolecular Nanoparticle (SMNP) Vectors
Our research lab has developed a self-assembled approach for preparation of a new category of nanoparticle vectors (a.k.a., Supramolecular Nanoparticles, SMNPs) for potential utilities as molecular diagnostics, therapeutics, and theranostics for cancer. The interplay of the β-cyclodextrin (CD) and adamantane (Ad)-based molecular recognition system enables a modular synthetic method for instantly producing a broad diversity of SMNP vectors with controllable sizes, surface chemistry, and payloads, via ratiometric mixing of three common molecular building blocks and payloads. Over the past 12 years, we have published a series of papers, demonstrating the feasibility of applying SNP vectors for a wide spectrum of diagnostic, therapeutic, and theranostic applications using cancer cell lines and animal models. Our current research efforts are devoted to exploiting the use of SMNP Vectors for CRISPR-Cas9-mediated genome editing to treat genetic diseases.
Dual Supramolecular Nanoparticle Vectors Enable CRISPR/Cas9‐Mediated Knockin of Retinoschisin 1 Gene—A Potential Nonviral Therapeutic Solution for X‐Linked Juvenile Retinoschisis
Supramolecular nanoparticle (SMNP) vectors were used for codelivery of two DNA plasmids—CRISPR‐Cas9 genome‐editing system and a therapeutic gene, Retinoschisin 1 (RS1)—enabling clustered regularly interspaced short palindromic repeats (CRISPR)‐associated protein 9 (CRISPR/Cas9) knockin of the RS1 gene with the homology‐independent targeted integration (HITI) strategy. The in vivo study involves intravitreally injecting the two SMNP vectors into the mouse eyes, followed by repeated ocular imaging by fundus camera and optical coherence tomography, and pathological and molecular analyses of the harvested retina tissues. Mice ocular organs retain their anatomical integrity, a single‐copy 3.0‐kb RS1/GFP gene is precisely integrated into the Rosa26 site in the retinas, and the integrated RS1/GFP gene is expressed in the retinas, demonstrating CRISPR/Cas9 knockin of RS1/GFP gene.
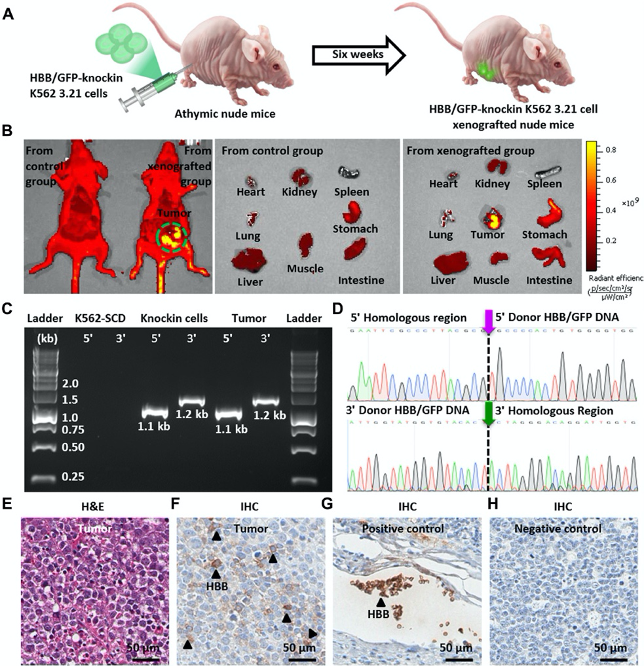
Supramolecular nanosubstrate–mediated delivery system enables CRISPR-Cas9 knockin of hemoglobin beta gene for hemoglobinopathies
Leveraging the endogenous homology-directed repair (HDR) pathway, the CRISPR-Cas9 gene-editing system can be applied to knock in a therapeutic gene at a designated site in the genome, offering a general therapeutic solution for treating genetic diseases such as hemoglobinopathies. Here, a combined supramolecular nanoparticle (SMNP)/supramolecular nanosubstrate–mediated delivery (SNSMD) strategy is used to facilitate CRISPR-Cas9 knockin of the hemoglobin beta (HBB) gene into the adeno-associated virus integration site 1 (AAVS1) safe-harbor site of an engineered K562 3.21 cell line harboring the sickle cell disease mutation. Through stepwise treatments of the two SMNP vectors encapsulating a Cas9•single-guide RNA (sgRNA) complex and an HBB/green fluorescent protein (GFP)–encoding plasmid, CRISPR-Cas9 knockin was successfully achieved via HDR. Last, the HBB/GFP-knockin K562 3.21 cells were introduced into mice via intraperitoneal injection to show their in vivo proliferative potential. This proof-of-concept demonstration paves the way for general gene therapeutic solutions for treating hemoglobinopathies.